More Information
Submitted: October 14, 2021 | Approved: October 21, 2021 | Published: October 22, 2021
How to cite this article: Jiang Y, Chen JH, Liu J, Wei T, Ge P, et al. The pathogenesis of psoriasis: insight into a complex “Mobius Loop” regulation process. Arch Pathol Clin Res. 2021; 5: 020-025.
DOI: 10.29328/journal.apcr.1001024
Copyright License: © 2021 Jiang Y, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Psoriasis; Pathogenesis; Mobius loop
Abbreviations: FDA: Food and Drug Administration; KC: Keratinocytes; AMPs: Antimicrobial Peptides; DC: Dendritic Cells; pDC: Plasma cell-like Dendritic Cells; NF-kb: Nuclear Factor Kappa-B; IL: Interleukin; TLR: Toll-Like Receptor; IFN: Interferon; MDC: Myeloid Dendritic Cells; TNF: Tumor Necrosis Factor; CD4+T: Cluster of Differentiation 4: CD8+T: Cluster of Differentiation 8; Th: T Helper Cell; ERK: Extracellular Signal-Regulated Kinase; TAK1: TGF-Β Activated Kinase 1; IKK: I-kb Kinase; AP-1: Activator Protein-1; ROS: Reactive Oxygen Species; CCL20: Chemokine (C-C Motif) Ligand 20; mTOR: Mammalian Target Of Rapamycin; EGFR: Epidermal Growth Factor Receptor; TGF-α: Transforming Growth Factor Α; DcR3: Decoy Receptor 3
The pathogenesis of psoriasis: insight into a complex “Mobius Loop” regulation process
Yuankuan Jiang1,2,4#, Haiyang Chen2-4#, Jiayue Liu2-4, Tianfu Wei2-4, Peng Ge2-4, Jialin Qu4* and Jingrong Lin1*
1Department of Dermatology, The First Affiliated Hospital of Dalian Medical University, No. 222, Zhongshan Road, Dalian 116011, China
2Institute (College) of Integrative Medicine, Dalian Medical University, No. 9, South Road of Lvshun, Dalian 116044, China
3Department of General Surgery, Pancreatic-Biliary Center, The First Affiliated Hospital of Dalian Medical University, No. 222, Zhongshan Road, Dalian 116011, China
4Laboratory of Integrative Medicine, The First Affiliated Hospital of Dalian Medical University, No. 222, Zhongshan Road, Dalian 116011, China
#Yuankuan Jiang and Haiyang Chen contributed equally to this work
*Address for Correspondence: Jingrong Lin, Department of Dermatology, The First Affiliated Hospital of Dalian Medical University, No. 222, Zhongshan Road, Dalian 116011, China, Email: [email protected]
Psoriasis is a chronic inflammatory skin disease with a complex mechanism, which is believed to be mainly based on immune disorders and activation of inflammatory pathways. However, we have combed through the literature and found that the pathogenesis of psoriasis might involve a “mobius loop” of “immunity-inflammation-oxidative stress-proliferation” process. The disordered immune environment of the skin might act as the basis, the outbreak of inflammatory factors as the mediator, and the imbalance of oxidative stress homeostasis as the activator. These factors work together, leading to abnormal proliferation of keratinocytes and further immune abnormalities, finally aggravating psoriasis. Therefore, here we review the latest evidence and advance in the pathogenesis of psoriasis, trying to contribute to further understanding and treatment of psoriasis.
Psoriasis is a chronic inflammatory skin disease with a complex mechanism associated with genetic factors and immune reactions. Although the global prevalence rate is about 2%, certain geographical differences are noticed. The incidence rate is low in Asia and some African regions, reaching 11% in Northern Europe and Caucasians [1-3]. At present, a radical cure of psoriasis is still unavailable, and there are nearly 6 million patients in China whose condition has not been effectively controlled [4]. In recent years, in-depth studies on the mechanism of psoriasis have pointed out new directions, providing new targets and strategies for further treatment of the disease.
In terms of modern medicine, in addition to the classical small-molecule drugs such as acitretin, methotrexate and cyclosporine, novel agents including apremilast and dimethyl fumarate have also been put into clinical application. In particular, the FDA approved biological agents such as secukinumab, Ixekizumab and Brodalumab, and put into clinical practice successively in the past five years [5-7]. Therefore, this article reviews the latest advances in the pathogenesis of psoriasis to provide a theoretical basis for treatment and drug selection of the disease.
The latest progress in the pathogenesis of psoriasis
Psoriasis is characterized by inflammatory infiltration, angiogenesis as well as abnormal proliferation and differentiation of keratinocytes. The pathogenesis is complicated. With the enrichment of genetics-immune-inflammation theory, more and more attention has been addressed to the loop regulation of oxidative stress and abnormal proliferation and differentiation of keratinocytes. We believed that the pathogenesis of psoriasis underwent the following three critical stages [8-10].
Under the background of genetic defects or abnormal modifications, inappropriate immune response provokes inflammatory reactions.
The inflammatory reactions are amplified in a cascade leading to a cytokine storm and then aggravated the imbalanced oxidative stress homeostasis.
Uncontrolled keratinocytes proliferation and dysfun-ctional differentiation are caused by inflammation. The proliferated keratinocytes participate in the vicious circle of “immune-inflammation-oxidative stress-proliferation-immune” leading to exacerbation and progression of psoriasis.
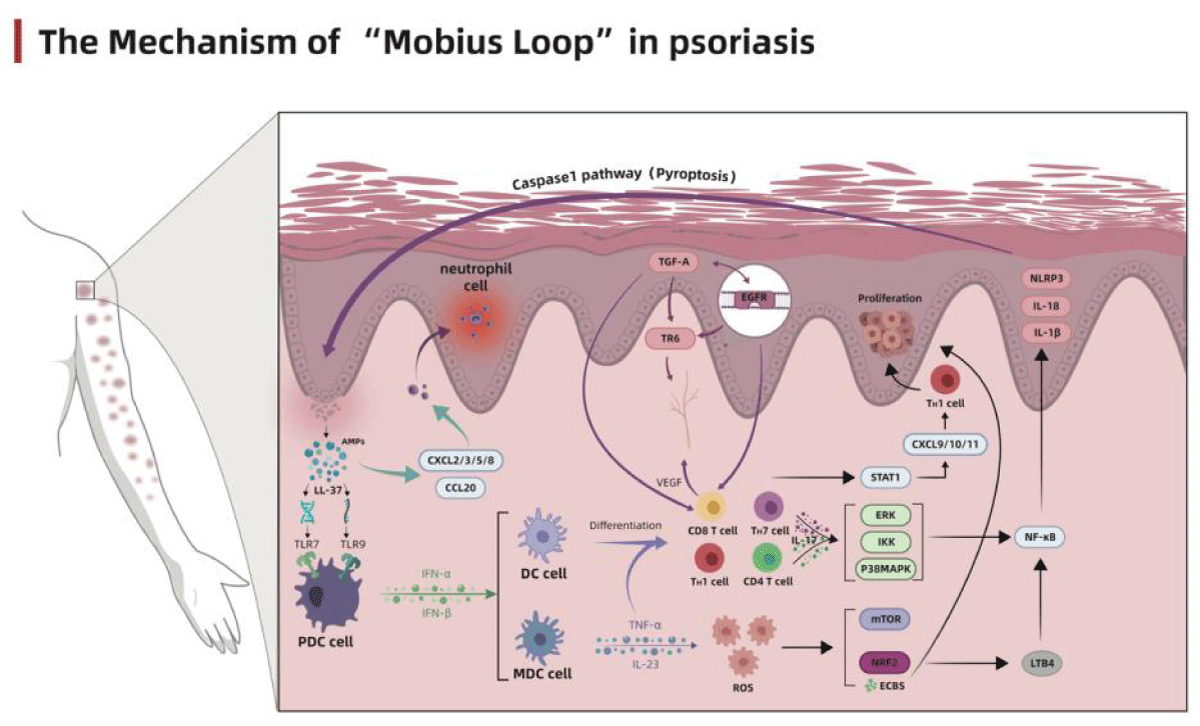
In this section, we will elaborate on the above vital stages respectively (Figure 1).
Figure 1: The Mechanism of “Mobius Loop” in Psoriasis.
Genetic and epigenetics factors
In recent years, the role of genetic factors in the pathogenesis of psoriasis has attracted increasing attention. A genome-wide study found that at least 60 chromosomal genes, including the PSORS1 gene, are susceptible sites for psoriasis [11,12]. The CARD14 locus, which constitutes the NF-κB scaffold protein, is mapped onto the PSORS1. The mutation at the CARD14 locus will cause abnormal activation of NF-κB and further amplify the inflammation in skin lesions [13]. In addition, IL-23, as a dimer structure composed of P40 and P19, has also exhibited multiple crossovers in the immune response of psoriasis. The corresponding IL-23R is also considered a high-risk gene for the development of psoriasis [14,15].
In addition to alterations of genetic sequence such as mutations and dislocations of susceptible genes, epigenetic changes may also trigger abnormal proliferation of keratinocytes and the release of inflammatory factors by activating target genes. The epigenetic mechanism in psoriasis mainly involves microRNA and methylation modification. On the one hand, studies on miRNA had revealed that miR-21, miR-31, miR-135b, miR-222 and miR-424 could induce the proliferation and differentiation of keratinocytes by promoting NF-κB activity [16-18]. Similarly, miR-203 could aggravate inflammation by enhancing transmembrane conduction via stimulating STAT3 signal, miR-210 could aggravate immune disorders by promoting the Th1 and Th17 differentiation through regulating T cell polarity [19-21]. On the other hand, methylation modification of genes may cause immune disorders by activating autoimmunity [22]. A clinical study found that the methylation level of m6A (an important anti-inflammatory factor that maintains immune homeostasis) in psoriatic lesions was significantly decreased compared with normal skin. At the same time, S100A9, which promotes cell proliferation and differentiation, was increased explosively [23-25].
The activation of genetic factors provides the incentive for the next immune disorder, while a series of activation of micRNA aggravates the inflammatory response and provides the basis for the pathological development of psoriasis.Therefore, although the genetic factors do not serve as independent pathogenic factors of psoriasis, they contribute to the immune-inflammation-proliferation process as an essential risk and additional factor.
Disorders in innate immunity and autoimmune response
The latest genome-wide association studies revealed that the disorders in innate immunity and autoimmune response are the major factors that induce the development of psoriasis and maintain the inflammatory environment [26]. Previous studies suggested that the prosiatic keratinocytes secret a series of antimicrobial peptides (AMPs), among which LL37 has attracted more attention. The LL37 released by keratinocytes binds with DNA and RNA, respectively, forming a complex that binds with the toll-like receptor 9 (TLR9) and TLR7 on the plasmacytoid dendritic cells (pDC), and stimulates the pDCs to secrete type I interferons (IFN-α and IFN-β) [27,28]. The secreted interferons will promote the maturation and differentiation of myeloid dendritic cells (mDC), Th1 and Th17 cells, and further accelerate cytokine secretion such as IFN-γ and IL-17 [29-31]. After maturation and activation, mDCs migrate to the lymph nodes and secrete tumor necrosis factor (TNF)-α, IL-23 and IL-12, promoting the proliferation and differentiation of Th17 and Th1 cells.
TNF-α and IL-23 activate the innate immune process by stimulating T cell subsets mainly composed of Th17 cells, thus maintain the inflammatory level of psoriatic lesions. In the meantime, the cytokines (IL-17, IL-21 and IL-22) secreted by T cells significantly promote the proliferation of keratinocytes [32]. Besides the activation of innate immunity, LL37 also acts as the autoantigen of T cells to initiate its autoimmune process. After LL37 presentation, the specific CD4+T cells secrete large amounts of IL-17, IL-21, IL-22 and IFN-γand aggravate the inflammatory reactions; the specific CD8+T cells are directly involved in epidermal growth, angiogenesis, presentation and activation process of other autoantigens such as keratin 17, and further promote the secretion of Th17-associated cytokines [33,34]. Therefore, the disorders of innate immunity and autoimmune response are the basis of the pathophysiological changes in psoriasis.
Inflammatory response and cytokine storm centered on the TNF-α–IL-23–Th17 pathway
As the immune environment changes, the pro-inflammatory molecules in the internal environment begin to express, inducing the infiltration of inflammatory cells, and triggering the inflammatory cascade. The TNF-α–IL-23–TH17 pathway plays a central regulatory role in this process. During the inflammatory cascade, TNF-α and IL-23 secreted by immune cells promote the maturation and differentiation of Th17 cells and secrete large amounts of IL-17. IL-17 is the central effector molecule of psoriasis [35], with the IL-17A exhibiting the strongest pro-inflammatory effect. A trimeric receptor complex formed by two IL-17RA subunits and one IL-17RC subunit could induce the recruitment of ACT1 adaptor protein and activate a large number of intracellular kinases, including extracellular signal-regulated kinase (ERK), p38 MAPK, TGF-β activated kinase 1(TAK1), and I-κB kinase (IKK) [36,37]. The abnormal activation of kinases will significantly activate transcription factors and chemokines such as NF-κB and AP-1, provoking a cytokine storm [38]. In addition to the regulatory mechanism of the TNF-α–IL-23–Th17 pathway, Th17 cells can also activate the STAT1 signaling pathway by secreting IL-26 and IL-29, and evoke a feedforward inflammatory response along with NF-κB to maintain the level of inflammation in the internal environment [38,39]. During the inflammatory cascade, the inflammatory signal was magnified successively, finally provoking a cytokine storm and recruiting many inflammatory cell infiltration to build a highly inflammatory extracellular environment and facilitate the abnormal proliferation and differentiation through breaking the oxidative stress balance.
The imbalance of oxidative stress homeostasis
During the immune and inflammatory stages, neutrophils in the blood proliferate massively and undergo oxidative burst by secreting large amounts of ROS under the stimulation of TNF-α [40]. ROS acts directly on keratinocytes and fibroblasts and causes a rapid decline in intracellular Ca2+ concentration, which could shift the state of cells from promoting differentiation to rapid proliferation [41,42]. It has been reported that increased ROS could increase mTOR secretion, thus directly activate NF-κB and stimulate the release of inflammatory cytokines[43]. With the accumulation of intracellular ROS, the intracellular antioxidant system was activated but insufficient to counterweigh the high levels of inflammation and oxidation in psoriasis. 0Instead, it increases secretion of K6, K16 and K17, promotes proliferation and inhibits apoptosis of keratinocytes, playing a protective role in hyperproliferative keratinocytes [44,45]. However, it is noteworthy that ROS functions more as a tandem role in the pathogenesis of psoriasis, which is initiated by abnormal immune environment and initiates the proliferation of keratinocytes through the metabolic.
Abnormal proliferation and differentiation of keratinocytes
Previous studies have shown that keratinocytes participate in the initial stage of a series of complex immune responses in psoriasis and act as the end effector cells that respond to the immune reactions, undergoing hyperproliferation and abnormal differentiation. After damage, the keratinocytes play the role of self-protection by releasing AMPs. However, in addition to the anti-infective effect of AMPs, they also activate the innate immunity, releasing large amounts of IL-17, IL-26, IL-29 and TNF-α to amplify the inflammatory reaction. In turn, the excessively released inflammatory factors act directly on keratinocytes, activate CXCL9/10/11 through the STAT1 pathway, and induce the Th1 cells to migrate into the epidermis [38,46].
In addition, keratinocytes can directly induce the activation of CCL20 and CXCL1/2/3/5/8 after the release of AMPs and promote the infiltration of immune cells such as neutrophils, macrophages and dendritic cells, facilitate the formation of Munro’s microabscess, and further promote the proliferation of keratinocytes to thicken the epidermis [38,47,48]. The existence of extensive parakeratotic cells in the stratum corneum enables sustained activation of the downstream regulatory mechanism of epidermal growth factor receptor (EGFR), leading to further proliferation and abnormal differentiation of keratinocytes [49]. With the increasing expression of EGFR, its ligand TGF-α is also overexpressed [50]. The specific combination of TGF-α with EGFR could amplify the signal sensitivity of TNF-α and IFN-γ to keratinocytes and aggravate the inflammatory reaction [51]. Meanwhile, sustained stimulation of high intensity by TNF-α will reversely activate through the NF-κB pathway[ 52], forming a loop regulation.
In addition, the sustained high level of EGFR also participates in angiogenesis via stimulating the expression of decoy receptor 3(DcR3/TR6) [53]. After NF-κB being abundantly expressed in keratinocytes, the expression of its downstream proteins, including NLRP3, Pro-IL-1β and Pro-IL-18 will also be accelerated. The precursors of the above proteins will be modified and activated by the activated caspase-1, not only hasten the proliferation of adjacent keratinocytes by activating pyroptosis and releasing large amounts of inflammatory factors, but also increase the polarity of T cells, induce their differentiation into Th17, and compel the keratinocytes into the vicious circle of immunity-inflammation-oxidation-proliferation-immunity, accelerating the progression of psoriasis[54-56].
It is widely known that the immune factor is undoubtedly a dominant factor in the pathogenesis of psoriasis. The inappropriate response to the immune system induces the inflammatory reaction, and the stepwise amplification of the signal leads to a cytokine storm, which stimulates the imbalance of oxidative stress homeostasis and aggravates the inflammatory response. The reinforced inflammatory reaction then acts on keratinocytes and dendritic cells, forming a closed-loop regulation. Keratinocytes play an essential role in this loop. They are the end effector cells of psoriasis undergoing hyperproliferation and abnormal differentiation under various inflammatory factors. The beginning of the loop regulation provokes the acquired immune pathway by releasing a series of antibacterial peptides LL37 after keratinocytes damage. We speculate that this might be the underlying cause of Koebner’s phenomenon in psoriasis. Therefore, the regulation mechanism of psoriasis is not a simple process of signal amplification step by step, but a complex “Mobius Ring” regulation process of immunity-inflammation-oxidation-proliferation-immunity by AMPs. Blocking the progress of this vicious circle has been considered the ideal treatment for various molecular drugs, biological agents, prescriptions and monomers from Chinese medicine. Unfortunately, no immune-inflammatory reaction could occur independently of the whole immune regulatory system, and excessive inhibition of any target on the pathway will interfere with other normal systems. For example, the application of biological agents specifically targeting IL-17 will also affect the intestinal inflammatory environment and cause the destruction of the intestinal barrier [57].
In the previous studies, keratinocytes were frequently used as the research object to investigate proliferation and changes of the inflammatory environment. Our literature review has revealed that the role of keratinocytes in immune regulation might be underestimated, and the keratinocytes regulatory loop might be an important treatment target. First, keratinocytes essential specificity as the essential cells of the skin. Targeting keratinocytes will not affect the immune homeostasis of other organs in the body. Second, the proliferation of keratinocytes is the primary clinical manifestation of psoriasis. Hyperproliferation of keratinocytes could be induced by infiltration of inflammatory factors and inhibition of their apoptosis as well. Autophagy is a well-recognized cytoprotective mechanism as opposed to apoptosis. However, it may not simply play a protective role in the proliferation and apoptosis of keratinocytes. The ROS accumulation caused by hypoxia condition due to keratinocytes proliferation and the consequent mitochondrial damage is a direct inducing factor for apoptosis [58].
In the keratinocytes of psoriasis, autophagy takes the initiative to clear the damaged cells and deceive the apoptosis monitoring system of the body when mitochondrial damage occurs. In addition, follicular phagocytosis is the process of digesting cells and their contents by lysosomes, mostly involving degradation at the protein level. Besides, the mRNA of accumulated inflammatory factors in damaged cells may diffuse to the external environment of keratinocytes through autophagy. Gene sequences in the functional domain may directly act on membrane receptors to activate antigen-presentation, as at the protein level, and further stimulate the proliferation and differentiation of keratinocytes [59,60]. Therefore, drugs such as camptothecin, which can not only inhibit cytokine storms, but also interfere with the autophagy and apoptosis of keratinocytes, will become a new option for the treatment of psoriasis [61-63]. Thus, we speculate that induction of keratinocytes into normally programmed apoptosis might also be an effective means of intervention in addition to an essential feature of psoriasis.
Author contribution
YJ and HC conceived this review and drafted the manuscript. JL and TW are responsible for the figure drawing and literature collation. PG and JL edited and finalized the manuscript for submission. JL and JQ reviewed and approved the submitted manuscript.
- Rachakonda TD, Schupp XW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014; 70: 512-516. PubMed: https://pubmed.ncbi.nlm.nih.gov/24388724/
- Christophers E. Psoriasis--epidemiology and clinical spectrum. Clin Exp Dermatol. 2001; 26: 314-320. PubMed: https://pubmed.ncbi.nlm.nih.gov/11422182/
- Parisi R, Symmons DP, Griffiths CE, Ashcroft DM, Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project team, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013; 133: 377-385. PubMed: https://pubmed.ncbi.nlm.nih.gov/23014338/
- Committee on Psoriasis, Chinese Society of Dermatology. Guideline for the diagnosis and treatment of psoriasis in China (2018 simplified edition) [J]. Chin J Dermatol. 2019; 52: 223-230.
- Gesser B, Johansen C, Rasmussen MK, Funding AT, Otkjaer K, et al. Dimethylfumarate specifically inhibits the mitogen and stress-activated kinases 1 and 2 (MSK1/2): possible role for its anti-psoriatic effect. J Invest Dermatol. 2007; 127: 2129-2137. PubMed: https://pubmed.ncbi.nlm.nih.gov/17495961/
- Lehmann JC, Listopad JJ, Rentzsch CU, Igney FH, von Bonin A, et al. Dimethylfumarate induces immunosuppression via glutathione depletion and subsequent induction of heme oxygenase 1. J Invest Dermatol. 2007; 127: 835-845. PubMed: https://pubmed.ncbi.nlm.nih.gov/17235328/
- Huang YW, Tsai TF. A drug safety evaluation of risankizumab for psoriasis, Expert opinion on drug safety. 2020; 19: 395-402. PubMed: https://pubmed.ncbi.nlm.nih.gov/32100591/
- Grän F, Kerstan A, Serfling E, Goebeler M, Muhammad K. Current Developments in the Immunology of Psoriasis. Yale J Biol Med. 2020; 93: 97-110. PubMed: https://pubmed.ncbi.nlm.nih.gov/32226340/
- Ni X, Lai Y. Keratinocyte: A trigger or an executor of psoriasis. J Leukoc Biol. 2020; 108: 485-491. PubMed: https://pubmed.ncbi.nlm.nih.gov/32170886/
- Christophers E, Metzler G, Röcken M. Bimodal immune activation in psoriasis. Br J Dermatol. 2014; 170: 59-65. PubMed: https://pubmed.ncbi.nlm.nih.gov/24117368/
- Bowcock AM, Krueger JG. Getting under the skin: the immunogenetics of psoriasis. Nat Rev Immunol. 2005; 5: 699-711. PubMed: https://pubmed.ncbi.nlm.nih.gov/16138103/
- Elder JT, Expanded Genome-Wide Association Study Meta-Analysis of Psoriasis Expands the Catalog of Common Psoriasis-Associated Variants. J Invest Dermatol. Symposium Proc. 2018; 19: 77-78. PubMed: https://pubmed.ncbi.nlm.nih.gov/30471756/
- Craiglow BG, Boyden LM, Hu R, Virtanen M, Su J, et al. CARD14-associated papulosquamous eruption: A spectrum including features of psoriasis and pityriasis rubra pilaris. J Am Acad Dermatol. 2018; 79: 487-494. PubMed: https://pubmed.ncbi.nlm.nih.gov/29477734/
- Di Meglio P, Di Cesare A, Laggner U, Chu CC, Napolitano L, et al. The IL23R R381Q gene variant protects against immune-mediated diseases by impairing IL-23-induced Th17 effector response in humans. PloS ONE. 2011; 6: e17160. PubMed: https://pubmed.ncbi.nlm.nih.gov/21364948/
- Kopp T, Riedl E, Bangert C, Bowman EP, Greisenegger E, et al. Clinical improvement in psoriasis with specific targeting of interleukin-23, Nature. 2015; 521: 222-226. PubMed: https://pubmed.ncbi.nlm.nih.gov/25754330/
- Guinea-Viniegra J, Jiménez M, Schonthaler HB, Navarro R, Delgado Y, et al. Targeting miR-21 to treat psoriasis. Sci Translatio Med. 2014; 6: 225re1. PubMed: https://pubmed.ncbi.nlm.nih.gov/24574341/
- Xu N, Meisgen F, Butler LM, Han G, Wang XJ, et al. MicroRNA-31 is overexpressed in psoriasis and modulates inflammatory cytokine and chemokine production in keratinocytes via targeting serine/threonine kinase 40. J Immunol. 2013; 190: 678-688. PubMed: https://pubmed.ncbi.nlm.nih.gov/23233723/
- Xu L, Leng H, Shi X, Ji J, Fu J, et al. MiR-155 promotes cell proliferation and inhibits apoptosis by PTEN signaling pathway in the psoriasis. Biomed Pharmacother. 2017; 90: 524-530. PubMed: https://pubmed.ncbi.nlm.nih.gov/28402921/
- Primo MN, Bak RO, Schibler B, Mikkelsen JG. Regulation of pro-inflammatory cytokines TNFα and IL24 by microRNA-203 in primary keratinocytes. Cytokine. 2012; 60: 741-748. PubMed: https://pubmed.ncbi.nlm.nih.gov/22917968/
- Wu R, Zeng J, Yuan J, Deng X, Huang Y, et al. MicroRNA-210 overexpression promotes psoriasis-like inflammation by inducing Th1 and Th17 cell differentiation. J Clin Invest. 2018; 128: 2551-2568. PubMed: https://pubmed.ncbi.nlm.nih.gov/29757188/
- Zhao M, Wang LT, Liang GP, Zhang P, Deng X, et al. Up-regulation of microRNA-210 induces immune dysfunction via targeting FOXP3 in CD4(+) T cells of psoriasis vulgaris. Clin Immunol. 2014; 150: 22-30. PubMed: https://pubmed.ncbi.nlm.nih.gov/24316592/
- Strickland FM, Richardson BC. Epigenetics in human autoimmunity. Epigenetics in autoimmunity - DNA methylation in systemic lupus erythematosus and beyond. Autoimmunity. 2008; 41: 278-286. PubMed: https://pubmed.ncbi.nlm.nih.gov/18432408/
- Suárez-Fariñas M, Lowes MA, Zaba LC, Krueger JG. Evaluation of the psoriasis transcriptome across different studies by gene set enrichment analysis (GSEA), PloS ONE. 2010; 5: e10247. PubMed: https://pubmed.ncbi.nlm.nih.gov/20422035/
- Li HB, Tong J, Zhu S, Batista PJ, Duffy EE, et al. m(6)A mRNA methylation controls T cell homeostasis by targeting the IL-7/STAT5/SOCS pathways. Nature. 2017; 548: 338-342. PubMed: https://pubmed.ncbi.nlm.nih.gov/28792938/
- Roberson ED, Liu Y, Ryan C, Joyce CE, Duan S, et al. A subset of methylated CpG sites differentiate psoriatic from normal skin. J Invest Dermatol. 2012; 132: 583-592. PubMed: https://pubmed.ncbi.nlm.nih.gov/22071477/
- Harden JL, Krueger JG, Bowcock AM. The immunogenetics of Psoriasis: A comprehensive review. J Autoimmun. 2015; 64: 66-73. PubMed: https://pubmed.ncbi.nlm.nih.gov/26215033/
- Morizane S, Gallo RL. Antimicrobial peptides in the pathogenesis of psoriasis. J Dermatol. 2012; 39: 225-230. PubMed: https://pubmed.ncbi.nlm.nih.gov/22352846/
- Morizane S, Yamasaki K, Mühleisen B, Kotol PF, Murakami M, et al. Cathelicidin antimicrobial peptide LL-37 in psoriasis enables keratinocyte reactivity against TLR9 ligands. J Invest Dermatol. 2012; 132: 135-143. PubMed: https://pubmed.ncbi.nlm.nih.gov/21850017/
- Nestle FO, Conrad C, Tun-Kyi A, Homey B, Gombert M, et al. Plasmacytoid predendritic cells initiate psoriasis through interferon-alpha production. J Experimen Med. 2005; 202: 135-143. PubMed: https://pubmed.ncbi.nlm.nih.gov/15998792/
- Gregorio J, Meller S, Conrad C, Di Nardo A, Homey B, et al. Plasmacytoid dendritic cells sense skin injury and promote wound healing through type I interferons. J Exp Med. 2010; 207: 2921-2930. PubMed: https://pubmed.ncbi.nlm.nih.gov/21115688/
- Santini SM, Lapenta C, Donati S, Spadaro F, Belardelli F, et al. Interferon-α-conditioned human monocytes combine a Th1-orienting attitude with the induction of autologous Th17 responses: role of IL-23 and IL-12, PloS One. 2011; 6: e17364. PubMed: https://pubmed.ncbi.nlm.nih.gov/21387004/
- Nestle FO, Turka LA, Nickoloff BJ. Characterization of dermal dendritic cells in psoriasis. Autostimulation of T lymphocytes and induction of Th1 type cytokines. J Clin Invest. 1994; 94: 202-209. PubMed: https://pubmed.ncbi.nlm.nih.gov/8040262/
- Yunusbaeva M, Valiev R, Bilalov F, Sultanova Z, Sharipova L, et al. Psoriasis patients demonstrate HLA-Cw*06:02 allele dosage-dependent T cell proliferation when treated with hair follicle-derived keratin 17 protein. Sci Rep. 2018; 8: 6098. PubMed: https://pubmed.ncbi.nlm.nih.gov/29666398/
- Fuentes-Duculan J, Bonifacio KM, Hawkes JE, Kunjravia N, Cueto I, et al. Autoantigens ADAMTSL5 and LL37 are significantly upregulated in active Psoriasis and localized with keratinocytes, dendritic cells and other leukocytes. Exp Dermatol. 2017; 26: 1075-1082. PubMed: https://pubmed.ncbi.nlm.nih.gov/28482118/
- Matsuzaki G, Umemura M. Interleukin-17 family cytokines in protective immunity against infections: role of hematopoietic cell-derived and non-hematopoietic cell-derived interleukin-17s. Microbiol Immunol. 2018; 62: 1-13. PubMed: https://pubmed.ncbi.nlm.nih.gov/29205464/
- Gaffen SL. Structure and signalling in the IL-17 receptor family, Nature reviews. Immunology. 2009; 9: 556-567. PubMed: https://pubmed.ncbi.nlm.nih.gov/19575028/
- Lee JS, Tato CM, Joyce-Shaikh B, Gulen MF, Cayatte C, et al. Interleukin-23-Independent IL-17 Production Regulates Intestinal Epithelial Permeability. Immunity. 2015; 43: 727-738. PubMed: https://pubmed.ncbi.nlm.nih.gov/26431948/
- Lande R, Botti E, Jandus C, Dojcinovic D, Fanelli G, et al. The antimicrobial peptide LL37 is a T-cell autoantigen in psoriasis. Nat Communicati. 2014; 5: 5621. PubMed: https://pubmed.ncbi.nlm.nih.gov/25470744/
- Hawkes JE, Chan TC, Krueger JG. Psoriasis pathogenesis and the development of novel targeted immune therapies. J Allergy Clin Immuno. 2017; 140: 645-653. PubMed: https://pubmed.ncbi.nlm.nih.gov/28887948/
- Naik HB, Natarajan B, Stansky E, Ahlman MA, Teagueet H, et al. Severity of Psoriasis Associates With Aortic Vascular Inflammation Detected by FDG PET/CT and Neutrophil Activation in a Prospective Observational Study. Arteriosclerosis, Thrombosis, Vascular Biol. 2015; 35: 2667-2676. PubMed: https://pubmed.ncbi.nlm.nih.gov/26449753/
- Magenta A, Dellambra E, Ciarapica R, Capogrossi mc. Oxidative stress, microRNAs and cytosolic calcium homeostasis. Cell Calcium. 2016; 60: 207-217. PubMed: https://pubmed.ncbi.nlm.nih.gov/27103406/
- Aksoy M, Kirmit A. Thiol/disulphide balance in patients with psoriasis, Postȩpy dermatologii i alergologii. 2020; 37: 52-55. PubMed: https://pubmed.ncbi.nlm.nih.gov/32467684/
- Young CN, Koepke JI, Terlecky LJ, Borkin MS, Boyd SL, et al. Reactive oxygen species in tumor necrosis factor-alpha-activated primary human keratinocytes: implications for psoriasis and inflammatory skin disease. J Invest Dermatol. 2008; 128: 2606-2614. PubMed: https://pubmed.ncbi.nlm.nih.gov/18463678/
- Ambrożewicz E, Wójcik P, Wroński A, Łuczaj W, Jastrząb A, et al. Pathophysiological Alterations of Redox Signaling and Endocannabinoid System in Granulocytes and Plasma of Psoriatic Patients, Cells. 2018; 7; 159. PubMed: https://pubmed.ncbi.nlm.nih.gov/30301214/
- Yang L, Fan X, Cui T, Wang G. Nrf2 Promotes Keratinocyte Proliferation in Psoriasis through Up-Regulation of Keratin 6, Keratin 16, and Keratin 17. J Invest Dermatol. 2017; 137: 2168-2176. PubMed: https://pubmed.ncbi.nlm.nih.gov/28576737/
- Wolk K, Witte K, Witte E, Raftery M, Kokolakis G, et al. IL-29 is produced by T(H)17 cells and mediates the cutaneous antiviral competence in psoriasis. Sci Transl Med. 2013; 5: 204ra129. PubMed: https://pubmed.ncbi.nlm.nih.gov/24068736/
- Harper EG, Guo C, Rizzo H, Lillis JV, Kurtz SE, et al. Th17 cytokines stimulate CCL20 expression in keratinocytes in vitro and in vivo: implications for psoriasis pathogenesis. J Invest Dermatol. 2009; 129: 2175-2183. PubMed: https://pubmed.ncbi.nlm.nih.gov/19295614/
- Homey B, Dieu-Nosjean MC, Wiesenborn A, Massacrier C, Pin JJ, et al. Up-regulation of macrophage inflammatory protein-3 alpha/CCL20 and CC chemokine receptor 6 in psoriasis. J Immunol. 2000; 164: 6621-6632. PubMed: https://pubmed.ncbi.nlm.nih.gov/10843722/
- Anderson KS, Petersson S, Wong J, Shubbar E, Lokko NN, et al. Elevation of serum epidermal growth factor and interleukin 1 receptor antagonist in active psoriasis vulgaris. Br J Dermatol. 2010; 163: 1085-1089. PubMed: https://pubmed.ncbi.nlm.nih.gov/20716221/
- Marinello E, Pastorelli D, Alaibac M. A case of psoriasis pustolosa palmaris induced by cetuximab. BMJ Case Rep. 2016; 2016: bcr2016214582. PubMed: https://pubmed.ncbi.nlm.nih.gov/26994052/
- Mascia F, Cataisson C, Lee TC, et al. EGFR regulates the expression of keratinocyte-derived granulocyte/macrophage colony-stimulating factor in vitro and in vivo. J Invest Dermatol. 2010; 130: 682-693. PubMed: https://pubmed.ncbi.nlm.nih.gov/19890352/
- Segawa R, Shigeeda K, Hatayama T, Threadgill D, Mariani V, et al. EGFR transactivation is involved in TNF-α-induced expression of thymic stromal lymphopoietin in human keratinocyte cell line. J Dermatol Sci. 2018; 89: 290-298.
- Mascia F, Mariani V, Girolomoni G, Pastore S. Blockade of the EGF receptor induces a deranged chemokine expression in keratinocytes leading to enhanced skin inflammation. Am J Pathol. 2003; 163: 303-312. PubMed: https://pubmed.ncbi.nlm.nih.gov/12819035/
- Swanson KV, Deng M, Ting JP. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol. 2019; 19: 477-489. PubMed: https://pubmed.ncbi.nlm.nih.gov/31036962/
- Cai Y, Xue F, Quan C, Qu M, Liu N, et al. A Critical Role of the IL-1β-IL-1R Signaling Pathway in Skin Inflammation and Psoriasis Pathogenesis. J Invest Dermatol. 2019; 139: 146-156. PubMed: https://pubmed.ncbi.nlm.nih.gov/30120937/
- Stamatopoulos A, Stamatopoulos T, Gamie Z, Kenanidis E, Ribeiro RD, et al. Mesenchymal stromal cells for bone sarcoma treatment: Roadmap to clinical practice. J Bone Oncol. 2019; 16: 100231. PubMed: https://pubmed.ncbi.nlm.nih.gov/30956944/
- Wang R, Li R. Adverse reactions of interleukin 17A and its receptor antagonist in the treatment of psoriasis (In Chinese). Chin J Dermatol. 2021; 54: 170-173.
- Mahmood A, Bisoyi P, Banerjee R, Yousuf M, Goswami SK. Mitoapocynin, a mitochondria targeted derivative of apocynin induces mitochondrial ROS generation and apoptosis in multiple cell types including cardiac myoblasts: a potential constraint to its therapeutic use. Mol Cell Biochem. 2021; 476: 2047-2059. PubMed: https://pubmed.ncbi.nlm.nih.gov/33515200/
- Lee HM, Shin DM, Yuk JM, Shi G, Choi DK, et al. Autophagy negatively regulates keratinocyte inflammatory responses via scaffolding protein p62/SQSTM1. J Immunol. 2011; 186: 1248-1258. PubMed: https://pubmed.ncbi.nlm.nih.gov/21160040/
- Qiang L, Yang S, Cui YH, He YY. Keratinocyte autophagy enables the activation of keratinocytes and fibroblasts and facilitates wound healing. Autophagy. 2020; 17: 2128-2143. PubMed: https://pubmed.ncbi.nlm.nih.gov/32866426/
- Lin J, Liu X, Hou S, An L, Lin X, et al. Effect of camptothecin on proliferation, Apoptosis and Telomerase Activity in HaCaT Cells (In Chinese). Chin J Dermatol Venereol. 2006; 10: 586-588.
- Guo Y, Zhang X, Wu T, Hu X, Su J, et al. Autophagy in Skin Diseases. Dermatology. 2019; 235: 380-389. PubMed: https://pubmed.ncbi.nlm.nih.gov/31269494/
- Hao Y, Zhang L. Effect of camptothecin on autophagy of HaCaT cells (In Chinese). Chin J Dermatol. 2017; 50: 86-90.